What is the Difference Between Lithium and Lithium-Ion Battery?

Lithium and lithium-ion batteries are two common types of batteries that are frequently used interchangeably. The performance characteristics of these two types of cells are quite similar i.e., they both offer on-the-go power, but they have a major difference that makes them suitable for different purposes.
A lithium-ion battery is often the kind that powers your phone or laptop; in contrast, a lithium cell can be used for various appliances that work with remote controls, etc. It is an older model that has been around for a long time. In this article, you will get insights into the major difference between lithium and lithium-ion batteries.
What is a lithium battery?

Lithium batteries have a long history of being used as a portable source of power for several electrical appliances. It is known for its primary cell construction which provides high power density and lightweight. However, it is not easy and safe to recharge them, so they are applicable for single use.
Lithium cells are available in a variety of sizes and voltages, but their chemical composition is kept unchanged. They are able to perform better for single-use as compared to Li-ion cells.
The manufacturing process is also a lot easier which makes them more cost-effective than other types of batteries. Furthermore, they are not easily degraded even after storing them for years. This is due to its chemical composition that still makes them useful for different things.
What is a lithium-ion battery?
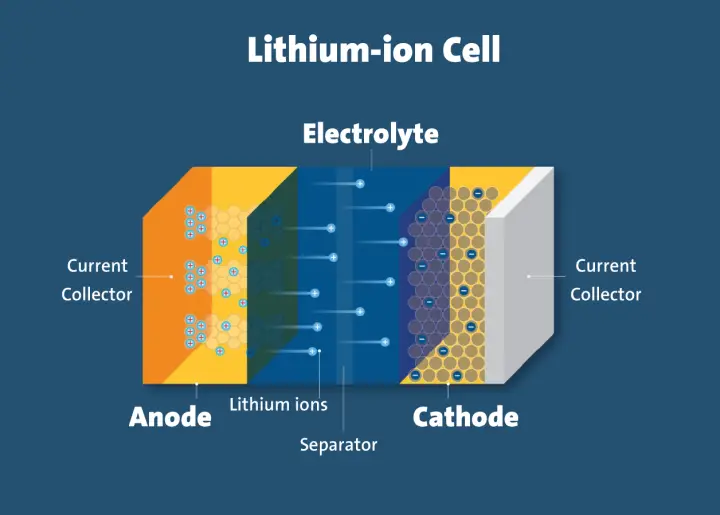
A lithium-ion battery is an upgraded version of lithium cells, which is known for its secondary cell construction that makes them easily rechargeable. These Li-ion cells consist of carbon cathode, lithium anode and electrolyte solution. The cathode is made from silicon or graphite which allows the flow of Li-ions into the cathode.
The reversible insertion of Li-ion into the cathode and anode structures makes it safe and easy to recharge over and over again until they are completely degraded. If you are looking for a rechargeable Li-ion cell then shop from EBL battery and buy good quality cells to power your electrical appliances at affordable rates.
Furthermore, it is the most common type of cell used nowadays to power various types of electrical appliances. It has multiple applications such as it is widely used in laptops, smartphones, tablets and various other electrical gadgets. The use of Li-ion cells is also used as a source of energy in electric vehicles.
The major distinction between lithium and lithium-ion batteries
1. Basic characteristics
The basic characteristics and features of the two types of batteries are one of the most significant differences between them. Lithium batteries are more powerful and lighter than Li-ion cells because they have a higher energy density. In addition to being more resilient and lasting longer than lithium-ion batteries, lithium cells are more powerful for single use.
Due to the durability of the Li-ion battery compared to its counterpart, can frequently be reused several times before needing to be replaced. It can supply more power for equipment with high power requirements, such as electric cars or computers because they have a greater voltage than lithium primary cells.
Moreover, a lithium battery is used to power a device for a limited amount of time that uses carbon as the electrolyte and lithium metal as the anode. A Li-ion cell can be used to power a device over and over again until it’s completely degraded.
2. Chemical composition
The positive and negative poles of a lithium battery are separated by a thin layer of lithium cobalt oxide. Graphite serves as the material for the negative pole while lithium cobalt oxide serves as the positive pole. A tiny coating of lithium cobalt oxide serves as the electrode/cathode, and a thicker layer of graphite serves as insulation.
A Li-ion battery consists of a substantial layer of graphite that serves as the insulator, and further layers of manganese dioxide (or other materials) are added to form an anode within the cathode structure. When current flows through the two electrodes during charging or discharging cycles, the electrolyte solution in this kind of battery enables ions to move between them.
3. Manufacturing and cost
The difference in the chemical composition makes the manufacturing process of lithium cells different from Li-ion cells, which leads to the difference in production cost and capacity. Furthermore, the frequency and other properties are also key factors in evaluating the overall cost of these batteries.
A lithium battery has a relatively low cost of production, but it also has a limited volume, making it challenging to create large quantities of them. A lithium-ion cell can be made in huge quantities all at once, despite having a relatively high production cost.
4. Applications and uses

Due to different chemical compositions, these two types of batteries have different applications and uses based on the device you are using and the amount of energy it requires to operate. Lithium batteries are not of much significance for day-to-day purposes due to the rechargeable properties of Li-ion cells, but it is often used in remote controllers and medical devices such as artificial cardiac pacemakers, etc.
A lithium-ion battery is a type of rechargeable cell that has more day-to-day purposes and is commonly used in smartphones, laptops, cameras, electric vehicles and other portable electronic gadgets. Also, it is frequently used for military and scientific applications to build weapons and spacecraft.
The Bottom-line
These were a few major differences between the two types of batteries commonly used in various modern electrical gadgets. Both cells have their pros and cons that make them ideal based on the purpose of your application.
Nowadays, people are using lithium-ion cells because they can be recharged multiple times and are environmentally friendly. However, it is also more convenient and easy to use. It is mostly favoured for modern electrical gadgets and has a long run-time. Most modern electrical gadgets can be powered with Li-ion batteries, so you don’t have to worry about changing the battery over and over again.
